Dmitri Mendeleev: The Father of the Periodic Table!
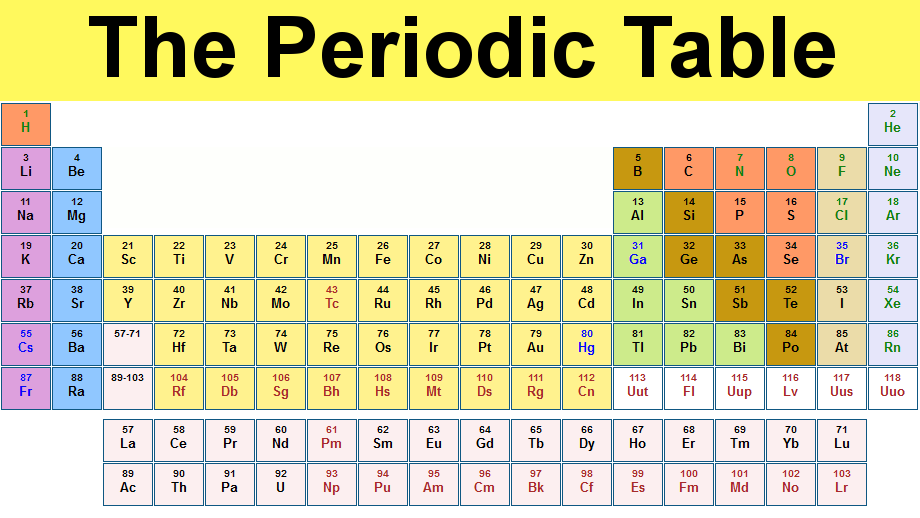
The Periodic Table
Groups of the Periodic Table
- Has 1 Valence Electron, Less Dense than other Metals, Highly Reactive, NOT found Naturally in Elemental Form, Clay-like, Can be CUT with a KNIFE, GOOD CONDUCTORS OF HEAT and ELECTRICITY- (Like ALL metals)
-Has 2 Valence Electrons, NOT found naturally in Elemental form, Silvery-White Color, Somewhat Reactive
-Varying Number of Valence Electrons- present in different shells of the nucleus- Readily form ALLOYS with themselves- -Brass-(copper and zinc), Conducts heat and Electricity, Forms Colored Compounds
-OTHER METALS- Ductile and Malleable, is different than transition elements, has high Density, and is Opaque.
-CHALCOGENS- Has 6 VALENCE ELECTRONS, from the greek language- CHALKOS, Mix of Metals, Metalloids, and non-metals.
-HALOGENS- Has 7 VALENCE ELECTRONS- Typically forms salt-table salt-(NaCi), is VERY reactive, is found in all three states of matter.
-NOBLE GASES- Has 8 VALENCE ELECTRONS, (Except Helium-1 or 2 valence electrons), For some Compounds, stable gases.
-The father of the periodic table is- Dmitri Mendeleev- took the elements and organized them by their properties and masses. Recognized that they had a repeating pattern as the masses increased.
HORIZONTAL ROWS- Periods VERTICAL COLUMNS- Groups/Family's
-Good conductors of electric current and heat. Solids at room temperature. Except for Mercury. Malleable- Hammered into thin sheets. Ductile- Drawn into thin wires. Form positive ions when bonding.
-Transition metals- Elements that form a bridge between the elements in the left and right sides of the table. Form positive ions when bonding.
-NONMETALS- Poor conductors of heat and electric current. Low bowling points, Many are gases at room temperature. --Solids at room temperature are brittle. Form negative ions when bonding- gain electrons.
-METALLOIDS- Elements with properties that fall between those metals and non-metals. Forms positive or negative ions when bonding. Good heat, poor electricity conductors.
-RARE EARTH METALS- Numbers 57-70, 89-102
-93 and above are Synthetic Elements. (Man-made)
-Alkaline Earth: MOST REACTIVE METALS
-Halogens: MOST REACTIVE NON-METALS
- Has 1 Valence Electron, Less Dense than other Metals, Highly Reactive, NOT found Naturally in Elemental Form, Clay-like, Can be CUT with a KNIFE, GOOD CONDUCTORS OF HEAT and ELECTRICITY- (Like ALL metals)
-Has 2 Valence Electrons, NOT found naturally in Elemental form, Silvery-White Color, Somewhat Reactive
-Varying Number of Valence Electrons- present in different shells of the nucleus- Readily form ALLOYS with themselves- -Brass-(copper and zinc), Conducts heat and Electricity, Forms Colored Compounds
-OTHER METALS- Ductile and Malleable, is different than transition elements, has high Density, and is Opaque.
-CHALCOGENS- Has 6 VALENCE ELECTRONS, from the greek language- CHALKOS, Mix of Metals, Metalloids, and non-metals.
-HALOGENS- Has 7 VALENCE ELECTRONS- Typically forms salt-table salt-(NaCi), is VERY reactive, is found in all three states of matter.
-NOBLE GASES- Has 8 VALENCE ELECTRONS, (Except Helium-1 or 2 valence electrons), For some Compounds, stable gases.
-The father of the periodic table is- Dmitri Mendeleev- took the elements and organized them by their properties and masses. Recognized that they had a repeating pattern as the masses increased.
HORIZONTAL ROWS- Periods VERTICAL COLUMNS- Groups/Family's
-Good conductors of electric current and heat. Solids at room temperature. Except for Mercury. Malleable- Hammered into thin sheets. Ductile- Drawn into thin wires. Form positive ions when bonding.
-Transition metals- Elements that form a bridge between the elements in the left and right sides of the table. Form positive ions when bonding.
-NONMETALS- Poor conductors of heat and electric current. Low bowling points, Many are gases at room temperature. --Solids at room temperature are brittle. Form negative ions when bonding- gain electrons.
-METALLOIDS- Elements with properties that fall between those metals and non-metals. Forms positive or negative ions when bonding. Good heat, poor electricity conductors.
-RARE EARTH METALS- Numbers 57-70, 89-102
-93 and above are Synthetic Elements. (Man-made)
-Alkaline Earth: MOST REACTIVE METALS
-Halogens: MOST REACTIVE NON-METALS
Picture sources for this page: http://www.chemicool.com/images/periodic-table.png, http://1.bp.blogspot.com/-j9tbz9TeVMI/UvQTgvFhNxI/AAAAAAAACyM/DyOx53zdiWQ/s1600/%D0%94%D0%BC%D0%B8%D1%82%D1%80%D0%B8%D0%B8%CC%86_%D0%98%D0%B2%D0%B0%D0%BD%D0%BE%D0%B2%D0%B8%D1%87_%D0%9C%D0%B5%D0%BD%D0%B4%D0%B5%D0%BB%D0%B5%D0%B5%D0%B2_4.gif,