Bonding and Naming
The part of an Atom that is responsible for bonding is: Electron
Ionic Bond- A bond involving 1 metal and 1 nonmetal element
Covalent Bond- A bond that involves NO metals
An atom that gives away its electrons in order to be stable has a positive charge.
An atom that takes electrons in order to be stable has a negative charge.
Chemical Bond- Forces that hold atoms or ions together as a unit
An Ionic Bond is a bond that holds cations and anions together.
Cation: positively charged ion Anion: negatively charged ion
METALS LIKE TO GIVE AWAY THEIR ELECTRONS NONMETALS LIKE TO TAKE ELECTRONS
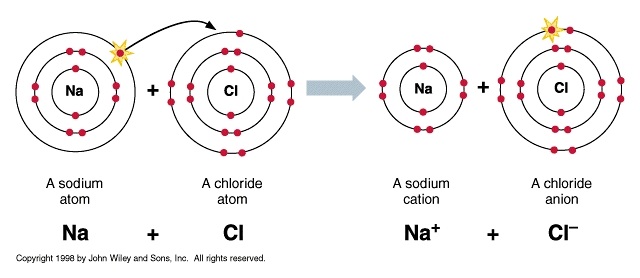
EXAMPLE- Sodium has one VALENCE ELECTRON. It likes to give away this ELECTRON to become HAPPY.
Chlorine has seven VALENCE ELECTRONS. It likes to take one electron to become happy.
Ionic Bond- A bond involving 1 metal and 1 nonmetal element
Covalent Bond- A bond that involves NO metals
An atom that gives away its electrons in order to be stable has a positive charge.
An atom that takes electrons in order to be stable has a negative charge.
Chemical Bond- Forces that hold atoms or ions together as a unit
An Ionic Bond is a bond that holds cations and anions together.
Cation: positively charged ion Anion: negatively charged ion
METALS LIKE TO GIVE AWAY THEIR ELECTRONS NONMETALS LIKE TO TAKE ELECTRONS
EXAMPLE- Sodium has one VALENCE ELECTRON. It likes to give away this ELECTRON to become HAPPY.
Chlorine has seven VALENCE ELECTRONS. It likes to take one electron to become happy.
PROPERTIES OF IONIC COMPOUNDS
-High Melting Point (801 `C)
-Poor Conductor when Solid
-Good Conductor when Melted
-Explained by - + attraction
-Dissolve in Water
-High Melting Point (801 `C)
-Poor Conductor when Solid
-Good Conductor when Melted
-Explained by - + attraction
-Dissolve in Water
|
Naming ionic compounds
First name: Metal name Last name: Nonmetal name but change the ending to ide - Halogens: change "ine" to "ide" - Nitrogen: Nitride - Oxygen: Oxide - Sulfur: Sulfide - Phosphorus: Phosphide If the metal has more than one possible charge First name: Metal name Middle name: Charge on the metal in Roman Numeral 1=I 2= II 3= III 4= IV 5= V 6= VI 7= VII 8= VIII Last name: nonmetal name but change the ending to ide Picture Sources: http://www.mrskscience.com/_/rsrc/1364668523142/chemistry-documents/bonding/james%20bond%20ionic%20bond.jpg?height=290&width=400 http://www.geo.arizona.edu/xtal/nats101/9_1.jpg quickmeme.com |